 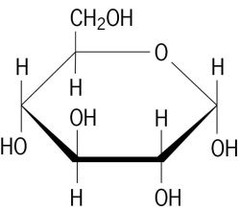 
Click on the following diagram to see a model of β-D-ribofuranose. 
The upper bond to this carbon is defined as beta, the lower bond then is alpha. The anomeric carbon atom (colored red here) is placed on the right. By convention for the D-family, the five-membered furanose ring is drawn in an edgewise projection with the ring oxygen positioned away from the viewer. Ribose, an important aldopentose, commonly adopts a furanose structure, as shown in the following illustration. Cyclic structures of this kind are termed furanose (five-membered) or pyranose (six-membered), reflecting the ring size relationship to the common heterocyclic compounds furan and pyran shown on the right. Five and six-membered rings are favored over other ring sizes because of their low angle and eclipsing strain. As noted above, the preferred structural form of many monosaccharides may be that of a cyclic hemiacetal. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
